
decarboxylation of two carbonate-bridged complexes of chromium(III) and. Chromium trichloride is also the starting material for many chromium (III) complexes. complexation of chromium(III) to NOM was intermediate between that of iron(III). It can be used as a raw material to produce many organic chromium compounds, such as diphenyl chromium, which is similar in structure to ferrocene. Anhydrous chromium trichloride is an important raw material in organometallic chemistry. There are spiral dislocations in the structure and do not contain metal-metal bonds. Chromium trichloride crystals contain CrCl6 octahedral units connected into layers. Chromium trichloride hexahydrate is a complex with three hydrated isomers: purple, light green and dark green solids. 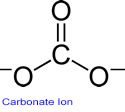
Chromium trichloride anhydrous is a strongly luminous purple crystal, almost insoluble in water. 
There are two kinds of anhydrous and hexahydrate. Information provided by: (external link)Ĭhromium trichloride (chemical formula: CrCl3) is a commonly used mordant and catalyst, easy to deliquesce, easy to sublimate, and easy to be oxidized by oxygen at high temperature. Chromium (VI) compounds are carcinogenic to humans (Group 1). There is sufficient evidence in experimental animals for the carcinogenicity of chromium (VI) compounds. Also positive associations have been observed between exposure to Chromium (VI) compounds and cancer of the nose and nasal sinuses. Chromium (VI) compounds cause cancer of the lung. There is sufficient evidence in humans for the carcinogenicity of chromium (VI) compounds. Lyon: International Agency for Research on Cancer. It is Solid in state and appears like Light Blue Solid At Room Temperature. Volume 100C: Arsenic, Metals, Fibres, and Dusts (PDF). Chromium(III) Carbonate is a chemical compound which can be represented using the chemical symbol Cr2(CO3)3. "chromitope sodium (Sodium Chromate, Cr 51) injection, solution". : CS1 maint: multiple names: authors list ( link) Chromium Carbonate is a water insoluble Chromium source that can easily be converted to other Chromium compounds, such as the oxide by heating (calcination). "A new method of potassium chromate production from chromite and KOH-KNO3-H2O binary submolten salt system". ^ Zhi Sun, Yi Zhang, Shi-Li Zheng, Yang Zhang (2009). Chromium(III) chloride Boiling Point 1300 C Molecular Weight 158.355 Appearance Purple shiny fluffy powder or flakes Safety 24/25 Risk Codes 22.Human exposure further encompasses impaired fertility, heritable genetic damage and harm to unborn children. The compound is also corrosive and exposure may produce severe eye damage or blindness. Sodium chromate is a strong oxidizer.Īs with other Cr(VI) compounds, sodium chromate is carcinogenic. In organic chemistry, sodium chromate is used as an oxidant, converting primary alcohols to carboxylic acids and secondary alcohols to ketones. It is a diagnostic pharmaceutical in determining red blood cell volume. Round your answer to the nearest percentage. This is the chemical formula for chromium (III) carbonate: Cr2 (CO3), Calculate the mass percent of oxygen in chromium (III) carbonate. It is also a dyeing auxiliary in the textile industry. Molar mass of Cr2 (CO3)3 284.019 g/mole Total atoms of O in one molecule of Cr2 (CO3)3 9. Na 2CrO 4 + H 2SO 4 → CrO 3 + Na 2SO 4 + H 2O Uses Īside from its central role in the production of chromium from its ores, sodium chromate is used as a corrosion inhibitor in the petroleum industry. 
So, we can determine that the formula for chromium (III) nitrate is Cr(NO3)3 (there is one. It converts to sodium dichromate when treated with acids:Ģ Na 2CrO 4 + 2HCl → Na 2Cr 2O 7 + 2NaCl + H 2Oįurther acidification affords chromium trioxide: All of the ion charges in an ionic substance always add up to zero. The industrial route to chromium(III) oxide involves reduction of sodium chromate with sulfur. Subsequent to its formation, the chromate salt is converted to sodium dichromate, the precursor to most chromium compounds and materials. For lab and small scale preparations a mixture of chromite ore, sodium hydroxide and sodium nitrate reacting at lower temperatures may be used (even 350 C in the corresponding potassium chromate system). Extensive studies on the solubility of amorphous. The process temperature is typically around 1100 ☌. Chromium(III)-carbonate reactions are expected to be important in managing high-level radioactive wastes. Typically calcium carbonate is included in the mixture to improve oxygen access and to keep silicon and aluminium impurities in an insoluble form. This process converts the chromium into a water-extractable form, leaving behind iron oxides. It is obtained on a vast scale by roasting chromium ores in air in the presence of sodium carbonate:ĢCr 2O 3 + 4 Na 2CO 3 + 3 O 2 → 4 Na 2CrO 4 + 4 CO 2 It is an intermediate in the extraction of chromium from its ores. It exists as a yellow hygroscopic solid, which can form tetra-, hexa-, and deca hydrates. Sodium chromate is the inorganic compound with the formula Na 2CrO 4.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
